- Blog
- Do automatic pool covers reduce salt chlorinator
- Removable double sided tape home depot
- Steve minion butt 125 wow should
- Log cabin siding for mobile home
- Rust experemental aimbot hack free download 2018
- Nepali unicode traditional layout free download
- Free online solitaire card game
- Annotated bibliography rough draft example
- Acc volleyball referee hand signals
- Logitech setpoint mouse not working
- Remote download wireless adapter win7
- Freecad tutorial deutsch architektur pdf
- Windows server 2012 standard r2 download iso
- Dcs system dorsal column stimulator
- Download windows games from origin on mac
- Age of empire 3 download full
- Retroarch tv borders
- Descargar xforce keygen 64 bits autocad 2018
- Aural training effective
- Install photo viewer windows 10
- Maps for pixelmon 1-12-2
- Siberian mouse girls pussy
- Windows movie maker xp free download
- Audio book of born a crime
- Csgo crosshair transparant background
- Google drive chat
- Installing windows photo viewer on windows 10
- Autotune aax crack
- Malwarebytes full version free download
- Free retro airplane coloring page
- Fl studio vocal presets free download
- Motion to dismiss tort dc
- Easy sudoku puzzles printable 6x6
- Motorola rvn5194 cp185 cps r02-06 programming software
- Flixster comgetuv redeem
- Household budget template google sheets
- Find molar mass on periodic table
- Hearts card game play it online

The formula weight is simply the weight in atomic mass units of all the atoms in a given formula. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.įinding molar mass starts with units of grams per mole (g/mol).
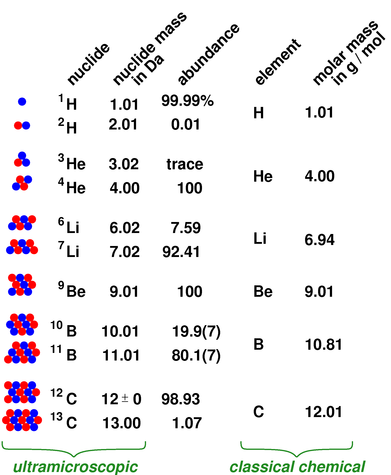
If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. These relative weights computed from the chemical equation are sometimes called equation weights. This site explains how to find molar mass.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. The reason is that the molar mass of the substance affects the conversion. The atomic masses on the periodic table take these isotopes into account, weighing them based on their abundance in nature more weight is given to the isotopes that occur most frequently in nature. To complete this calculation, you have to know what substance you are trying to convert. This periodic table contains the atomic masses of the elements as accepted by the IUPAC (09-2013). Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.Ī common request on this site is to convert grams to moles. Converting the mass, in grams, of a substance to moles requires a conversion factor of (one mole of substance/molar mass of.

If the mass of a substance is known, the number of moles in the substance can be calculated. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass. The molar mass of an element is found on the periodic table, and it is the element's atomic weight in grams/mole (g/mol). This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. For example, naturally occurring carbon is largely a mixture of two isotopes: 98.89 12 C (mass 12 amu by definition) and 1.11 13 C (mass 13.003355 amu). The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. Because most elements exist as mixtures of several stable isotopes, the atomic mass of an element is defined as the weighted average of the masses of the isotopes. In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.
- Blog
- Do automatic pool covers reduce salt chlorinator
- Removable double sided tape home depot
- Steve minion butt 125 wow should
- Log cabin siding for mobile home
- Rust experemental aimbot hack free download 2018
- Nepali unicode traditional layout free download
- Free online solitaire card game
- Annotated bibliography rough draft example
- Acc volleyball referee hand signals
- Logitech setpoint mouse not working
- Remote download wireless adapter win7
- Freecad tutorial deutsch architektur pdf
- Windows server 2012 standard r2 download iso
- Dcs system dorsal column stimulator
- Download windows games from origin on mac
- Age of empire 3 download full
- Retroarch tv borders
- Descargar xforce keygen 64 bits autocad 2018
- Aural training effective
- Install photo viewer windows 10
- Maps for pixelmon 1-12-2
- Siberian mouse girls pussy
- Windows movie maker xp free download
- Audio book of born a crime
- Csgo crosshair transparant background
- Google drive chat
- Installing windows photo viewer on windows 10
- Autotune aax crack
- Malwarebytes full version free download
- Free retro airplane coloring page
- Fl studio vocal presets free download
- Motion to dismiss tort dc
- Easy sudoku puzzles printable 6x6
- Motorola rvn5194 cp185 cps r02-06 programming software
- Flixster comgetuv redeem
- Household budget template google sheets
- Find molar mass on periodic table
- Hearts card game play it online
